epHects
Frank
Partner name: Mariana Nunn
Purpose: The purpose of this lab experiment is to further explore the effects of ocean acidification and changing pH’s of water.
Procedure: In order to complete this experiment, we placed various shells into a few substances with different pH’s. After a period of time, we re-evaluate the shells, and see the effects.
Pre-Lab Questions:
Partner name: Mariana Nunn
Purpose: The purpose of this lab experiment is to further explore the effects of ocean acidification and changing pH’s of water.
Procedure: In order to complete this experiment, we placed various shells into a few substances with different pH’s. After a period of time, we re-evaluate the shells, and see the effects.
Pre-Lab Questions:
- What is pH?: pH measures the acidity of a substance like water. It is on a scale from 0-14
- What does pH stand for?: pH stands for potential hydrogen
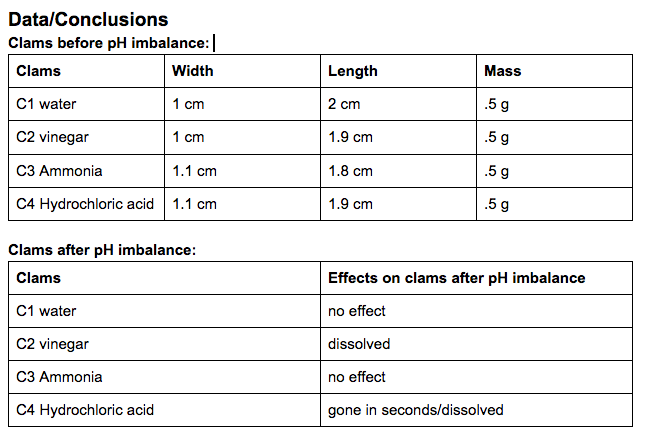
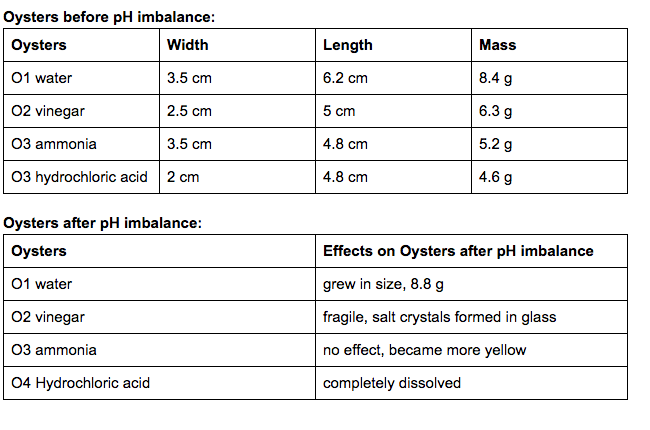
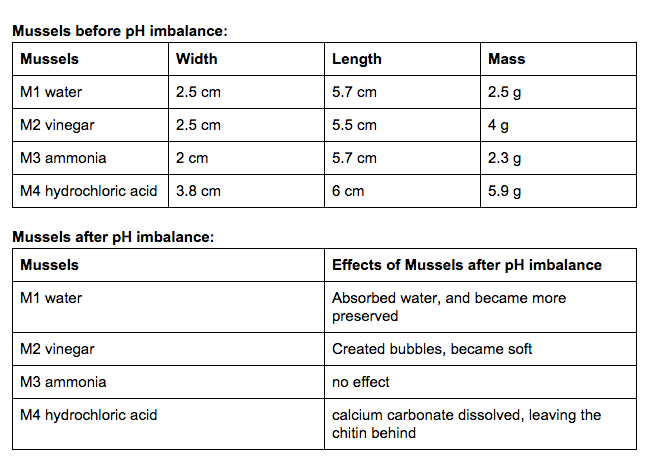
Summary of observations: The water had very minimal to no effect on the shells. All of them were generally similar to when we first put them in. The same can be pretty much said about the ammonia cleaner. The ammonia pretty much caused a slight discoloration, and that is about it. The vinegar on the other hand caused a lot of deterioration among the shells. It caused the clam to completely disappear, and made a lot of the oyster deteriorate. The calcium carbonate of the mussel dissolved leaving behind the chitin. The hydrochloric acid did a quick job of almost completely disintegrating everything in a matter of minutes.
Conclusion: When things turn acidic, they have effects that do not translate to healthy nature. If the ocean acidification speeds up, we may be in a lot of trouble soon.